ScienceKindergartners41 Comments
Set up a mega science experiment to figure out what baking soda and vinegar ratio works best for you and your preschooler!
We love a good baking soda and vinegar experiment, don’t you!??
I don’t know how many times the kids and I have done experiments with baking soda and vinegar.
But I’ve always questioned how much to use of each…
I’ve always just guessed and it turned out however it turned out.
Sometimes it was a great fizzy explosion, other times it just plain fizzled out.
However, the boys have always had a good time with our experiments.
But I thought it was time to figure out the ratio to make the best fizzy “explosion” using just baking soda and vinegar.
And letting the kids decide what worked the best.

First of all, why do baking soda and vinegar react the way they do?
I’m not all science-y and understand chemical reactions, so I had to look up a good answer for you from Steve Spangler Science.
The bubbles and foam you see are filled with carbon dioxide gas (CO2) that’s being released by an acid/base reaction. Vinegar is acetic acid dissolved in water and baking soda is a base called sodium bicarbonate. Initially, the reaction makes carbonic acid which is unstable. It quickly breaks down into CO2 and water. The gas then rapidly leaves the water creating foam and bubbles along the way.
You can read more about how the science behind this experiment as well as learn how to make and amazing rocket propelled by baking soda and vinegar over on Steve Spangler Science.
What ratio of baking soda to vinegar is best?
I set up an experiment for Henry and I to discover how to get the best baking soda and vinegar results.
We kept it a simple mixture, with just these 2 ingredients:
- baking soda
- vinegar
No added food coloring to worry about stains or anything like that. Keeping it very simple.
It might be handy to set this up on a tray, or outside and keep a sponge or towel handy to clean up the fizzy overflow.
I set up 3 glasses (clear).
Then, I had Henry write the numbers 1-2-3 each on a piece of paper to label the glasses so he could write down the results of his experiment.
The More Vinegar Experiment
First, I started by pouring vinegar in each glass.
I corresponded the number of tablespoons of vinegar to the number on Henry’s papers.
- 1 tablespoon
2 tablespoons - 3 tablespoons

For this initial experiment, we used one tablespoon of baking soda as our constant for this round of experimenting.
Henry measured one tablespoon of baking soda and dumped it into the first glass with one tablespoon of vinegar and observed what happened.
We kept doing this (always with one tablespoon of baking soda) through all three glasses and found that the fizzing got bigger and better with each additional tablespoon of vinegar.
Another round of testing would be done to see the reaction with even more vinegar!
Does Even More Vinegar Get a Better Fizz?
Another setup was done with more vinegar, to see if the explosions would keep getting biger.
This time we tried more vinegar in each glass:
- 4 tablespoons
- 5 tablespoons
- 6 tablespoons
Each still with one tablespoon of baking soda poured in to test.
We found once again that the fizzing was quite a bit bigger and better with 6 tablespoons as a result.
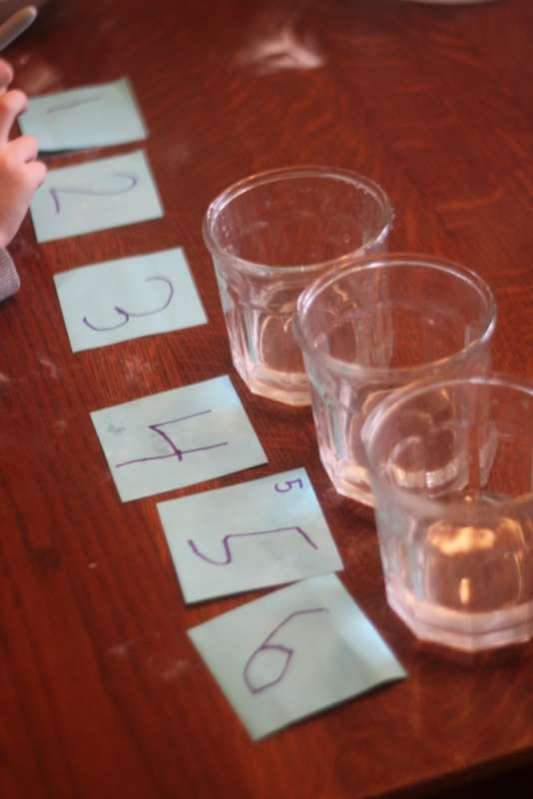
Yes! More vinegar equals more fizz!
Yet another round of testing needed to be done. This time I skipped numbers and didn’t do consecutive counting so we could really see the difference.
I set up the cups each with different amounts of vinegar:
- 3 tablespoons
- 6 tablespoons
- 12 tablespoons
Whoa! 12 tablespoons of vinegar to 1 tablespoon of baking soda overflowed our glasses!
(Told you the sponge may come in handy for cleanup! However, baking soda and vinegar make excellent detergents, so maybe you could do this in the bathtub and scrub the soap scum off in the process! Ha!)

I can’t help but wonder if we were to try it with even more vinegar what the result would be.
But our glass was overflowing the way it was, so that was the end of our experimenting to find out the best ratio.
The next question I had…
Is it better to add baking soda to vinegar? Or vinegar to baking soda?
Would twelve tablespoons of vinegar have the same reaction if the roles were reversed and we added it to one tablespoon of baking soda, or does it not matter?
Would the chemical reactions be the same?
We HAD to find out!

So we tested that as well with the best ratio of baking soda to vinegar above.
The amount of fizzing was the same for both.
However… there is a difference in the time of reaction between the two.
When you add baking soda to vinegar like we did the first experiment above, the reaction it creates is sort of delayed, building up to a big fizz. But it’s slow building.
On the flip side, when you flip flop the roles and add the vinegar to the baking soda , the reaction is immediate and almost explosive.
Depending on the experiment you’re doing, you may want to do it one way over the other.
Our Mega Baking Soda and Vinegar Experiment Results:
Adding vinegar to baking soda gives you an immediate reaction. Adding baking soda to vinegar, the reaction is delayed, but then fizzes the same amount.
More vinegar is better. A 12 to 1 ratio of vinegar to baking soda caused a fizzing explosion!
Who knew!?
We could have kept going with this all afternoon! Henry was getting a kick out of the experiment and loved watching it overflow the cup.
Do you add baking soda to vinegar or vinegar to baking soda when you do these experiments?
When we do our blow up a balloon experiment, it is adding the baking soda to the vinegar that is already in the bottle. Which is a delayed fizz. I wonder what would happen if we did the opposite and reversed the roles? Would the balloon blow up more rapidly? Would the balloon explode?
That sounds like an experiment we need to try, or you need to try and report back and tell me how it turns out!
What experiments have you done with vinegar and baking soda?

WANT TO SAVE THIS ACTIVITY?
Enter your email below & we'll send it straight to your inbox so you can access this activity later! Plus, you'll get simple activities from us every week!
 Baking Soda and Vinegar Experiment with Color Fun
Baking Soda and Vinegar Experiment with Color Fun
 10 Tips for Cleaning Up the Mess After Fun Activities
10 Tips for Cleaning Up the Mess After Fun Activities Discover an Easy Magical Color Changing Milk Experiment for Kids
Discover an Easy Magical Color Changing Milk Experiment for Kids Easy Homemade Fizzy Sidewalk Paint Recipe for Kids
Easy Homemade Fizzy Sidewalk Paint Recipe for Kids










Alex says
Has anyone ever tried adding more baking soda to the ratio? Like what if once you got to 12 tablespoons of vinegar you added more baking soda?
Tabitha Lightfoot says
Great idea! We didn’t try that! If you try it, let us know how it goes!
Student says
What if you add more baking soda?
ritchy says
good job keep it up
Jahseh says
Try sulfuric acid more reaction
lisa m fournier says
I would have loved to know the differentials between different types of vinegars being added to the baking soda.
Tabitha Lightfoot says
Hi Lisa! It was always just normal vinegar – the part that was different was the amount that was added to each cup.
Peter says
Another thought … has anyone determined the volumes of ingredients that work best in various bottle sizes? For instance, my Library class will be using 20oz bottles. Some use 16. Some use liter. If the bottle is filled too full, insufficient amounts of CO2 will form, reducing the height of the rocket. Not enough mixture volume and no lift off.
Peter says
I love the trial and error nature of this. Plus you don’t have to actually launch to see the results of this portion of the experiment. I think I’ll try and go beyond the 12:1 ratio and see what I can determine. Might be difficult to measure. Thoughts?
Tabitha Lightfoot says
Hi Peter – we haven’t tried anything beyond 12:1 to see what happens but if you did it in a larger glass, you could definitely try it out! And, experimenting with different size bottles is a great idea! Let us know how it goes if you try it out!
chantol says
how do you measure the eruption?
Tabitha Lightfoot says
Hi Chantol! Great question – for this one, they just watched the reactions to see how high the fizz went in the glass.
Timmer says
Thanks so much for posting your experiment! – Not only did this answer a question I had, but it’s a wonderful example of exploring along with your kids! They learn a lot from watching how YOU go about asking and answering questions.
Amy Hopkins says
Hi there! I’m a TK/K teacher and will be inviting the first-/second-grade class to join us for my very first vinegar/baking soda experiment in the classroom. Your informational website was very helpful! Thank you for sharing your experiment test results! The students are going to be so excited!!!
Sincerely,
Amy Hopkins
Steve Hoover says
I arrived at a different ratio for actually making CO2. I used a 12 oz jar and measured how much CO2 was in the jar by lowering a lit match in it and watching the level it went out at. I extracted gas from the jar with an animal feeding syringe I got from Tractor Supply and counted how many extractions it took to empty the jar based on match tests. I started with 3 to 1 which filled the jar and took 10 extractions to empty it. I don’t know if it overflowed the jar. Then I tried the 12 to 1 which only half filled the jar and at 5 extractions it was empty. Then I tried 6 to 1 and again was able to get 10 extractions. So IMO 1/2 teaspoon to 1 tablespoon, 6 to 1 is the ideal for making CO2.
Luke says
I am doing that for my science project in my secondary school. : )
K says
Bake Soda & Vinegar. I just found my intro experiment to review Scientific Method for my HS Honors Chem class. Thanks! LOL.
Fay Rowland says
brillo, thanks, I’ve always wondered what the best ratio is, many thanks xx
Apple says
Why vinegar and baking soda mixed will exploded?
Jyoti says
I am studying in class 7. I had got a project of showing this experiment in class. Thanks for giving me tips about it.
ola says
Because vinegar is a base while baking soda is an acid. When acid and base are mix together they will react.
Thank you.
Chemist says
Ideal ratio is 70g/L. A tablespoon is 15g and so will react with up to 215mL or around 14 tablespoons. :)
savannah bussey says
Thanks for the great idea for the project. instead i used ballons over the top! But it was a amazing projext!! :)
Andi says
if you add a drop of dish detergent to your vinegar it will make the eruption last longer.
Jamie Reimer says
No way!!!! I did NOT know that Andi! Thank you so much for the tip! I’ll be trying that next time for sure!
maryanne @ mama smiles says
I love the way you turned this into a full-blown science experiment!
Jamie Reimer says
Thanks Mary Anne! :) It was so much fun!
Daisy Broomfield says
What great science and what a useful thing to know! We often set off a volcano with baking soda and vinegar and have generally added more powder when it stopped fizzing- your test suggests we should use more vinegar. Thanks for the tip (I think my boys would like to do the research themselves too- just to check!!)
Jamie Reimer says
Yeah I think vinegar would get more out of it…. but maybe not either if its the second time around… because the soda would be dissolved… I’m not sure! Another thing to test!